Acadia Pharmaceuticals (#ACAD) is set to release their top-line results from the -020 clinical trial of their lead drug Pimavanserin for treatment of Parkinson's disease psychosis (PDP). In a press release just issued, the company announced that they will be hosting a conference call to discuss top-line results tomorrow at 8AM EST.
Earlier today we were looking at the ACAD 5-day 5-minute chart and saw an interesting pattern:
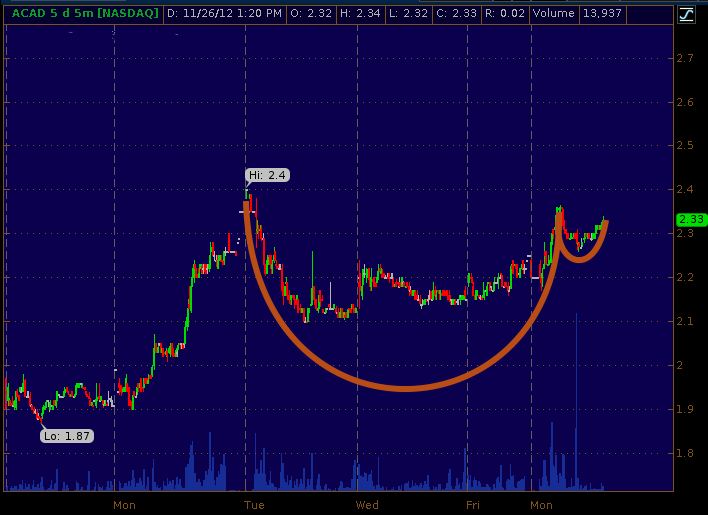
Notice the cup-and-handle pattern that was beginning to form on the chart, indicating that a move to the upside may occur in the near future.
We did not publish this chart at the time because chart patterns like this are not very reliable on such short timescales. Many factors are at work in intra-day movements and what looks like a true cup and handle at 1pm may turn out to be garbage by 2pm.
A recent article suggested that fair value for ACAD ahead of the clinical trial data release was around $2.64 per share. The chart above certainly supports a return to this level; however, whether or not traders would give the stock such a valuation ahead of the data release remained an open question at that time.
Here is the updated chart including the rest of the trading day and the after-hours reaction to the press release:
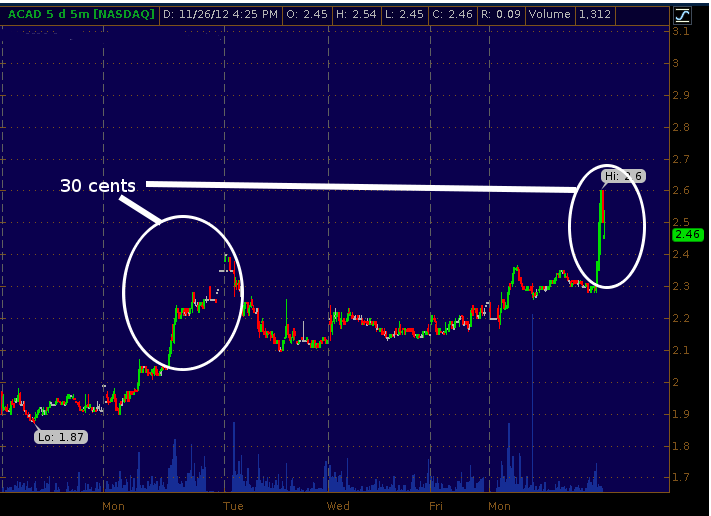
Notice that immediately after the press release, a 30 cent move occurred. This is exactly the move that was predicted by the cup and handle pattern in the first chart above, and the $2.60 price point is about where fair value for ACAD should be ahead of the data.
As we have outlined previously, we believe that the re-designed clinical trial protocol bodes well for the -020 study, but as seasoned biotech traders know, nothing can be taken for granted in this space. The way the company is going about releasing the data has been taken to be bullish by the market. The press release (linked above) simply announces that the data will be released tomorrow. In effect, the company is building anticipation for the data release. There is no good reason to build anticipation if the news your are about to release is bad. It's always better to get bad news out without fanfare; therefore, the announcement of the timing of a conference call without the announcement of the actual results has been read as largely positive.
As biotech blogger Adam Feuerstein noted on stocktwits, some biotech companies in the past (specifically Pionard Pharmaceuticals) have pre-scheduled conference calls about clinical trial data only to announce trial failure. He also mentions however that ACAD did NOT have a pre-scheduled conference call ahead of their 2009 clinical trial data release for the -012 study (which failed to meet its primary efficacy endpoint).
Recall that the -020 study for ACAD was designed after the company data-mined the -012 study to find that the sicker patients (moderate to severe PDP) showed a strong response to the highest dose (40mg/day) of Pimavanserin, and that the 9-items of the Scale for the Assessment of Positive Symptoms (SAPS) that specifically related to psychosis showed a statistically significant clinical benefit to Pimavanserin over placebo. Such post-hoc analysis can be subject to bias which is why FDA does not simply approve drugs based on a-posteriori hypothesis testing from trial data. If the -020 study results are positive, this is a huge win for ACAD which currently has no other viable drug candidates in advanced clinical trials.
For ACAD, these trial results are truly a binary event. The stock is worth $5 or more on positive results, and worth no more than available cash (~35 cents per share) on negative news. Based on the expected timing of the data release this week, we suggested to our members this morning that scaling back their long exposure to ACAD stock would be a prudent move if they did not want to hold through the binary event. For our Red Acre Partners L.P. Micro-Cap Value Fund, we moved half our holdings from shares to calls today, thereby reducing our cost basis while still being able to participate in any potential upside. Having been ahead of the curve on a large move in ACAD's price earlier this year, our current holdings are more or less "house money" which is the only reason we'll hold through the binary event.
What to Expect After the Data Release
On positive news, the upward move of the stock will be immediate, but, if the stock gets ahead of itself and goes much beyond $5, a subsequent run-down in price will occur over the ensuing weeks. Watch the stock price after the data is released to see whether or not there is an overly bullish reaction to the news. On a strong move above $5.50 a short position, or selling of calls dated at least 3 months out is likely to be a winning trade. Once the euphoria of positive news settles, the market will realize that the company still has to do a second phase III clinical trial (the -021 study) before Pimavanserin can be approved. Given the 18+ months it will take to do this confirmatory trial, the stock is likely to settle back in to a realistic valuation after any initial overbuying.
On the other hand, negative trial results, or, for that matter, equivocal trial results, will send ACAD into a tailspin. If this occurs and you held long in hopes of a positive result, cut your loses and move on. There are plenty of other binary event trades out there even between now and the end of the year.